A new cancer therapy with high on-target efficacy and minimal side effects
Talking with Professor Simon Barry on cancer immunotherapy …
Q: Simon, you are a hospital scientist at the Women’s and Children’s Hospital in Adelaide and professorial fellow in the Robinson Research institute, part of the University of Adelaide. What has led you to researching immunotherapy?
A: I started out doing a Bachelor of Science degree in Biochemistry, which is the mixture of how biology and chemistry talk to each other, and then went on to do a doctoral degree in a kind of immunology looking at the question of “how does a particular cell get born and get programmed?”. Since then I have developed an interest in “how does the immune system work and how does it recognise things that it should see, such as infections or viruses, but be smart enough not to mistake things as threats that it shouldn’t (like your normal tissues and food in your gut etc.)?”.
In the 90’s this was poorly understood; then in the 2000’s I was working for a biotech company very interested in the area of auto-immune illnesses (e.g. arthritis), and controlling the immune system … that’s when I got very interested in what normal or abnormal immune responses look like … leading me to this area of cancer immunology.
Q: Immunotherapy is a new treatment many may not have heard of, and if people have heard of it many do not know what it means. Can you explain it in layman’s terms?
A: Immunotherapy is an umbrella term that captures “a way that the body harnesses the immune system and makes it good to recognise cancer cells in the body”. There is more than one immunotherapy and it’s been evolving fast. Early clinical science in immunotherapy about 10 years ago was using a process of taking a biopsy of some immune system cells that had recognised an existing tumour, then ‘grow those cells’ (i.e. those cells helping to fight the cancer). They would then deliver the cells back to the person. There was a lot of excitement about this process, but this turned out not to hold the great promise we all hoped for.
Recently, it became more obvious that the immune system is trying to recognise cancer in anybody, but it can fail because it is getting confused by the fact that cancer cells are “sort of a normal tissue” and that’s the reason why your own immune system doesn’t immediately recognise it and reject it as cancer. This understanding has led to the next wave of immunotherapy, approaching the question “how does cancer ‘turn off’ the immune system?”. It turns out that the cancer cells have learnt how to ‘talk to the immune system’ and ‘hypnotise’ it.
This next wave of immunotherapy, which is hitting the clinics now, is driven by understanding how the ‘hypnosis’ works and then using antibodies to ‘block the hypnosis’. These antibody therapies are known as ‘check point inhibitors’ and they are having big successes in the last 3-5 years in some cancers, but not every cancer type. Cancer cells are good at determining what works and what doesn’t. This means they change themselves so that the check point inhibitors may no longer be effective. It is in some cases. It’s similar to the flu virus, you know how we need a different vaccine every year. Cancer cells can learn how to give the immune system a new “leave me alone” signal. This has led to the most recent phase, the CAR T-cell approach which is what I’m working on.
So the short history of Immunotherapy is that check point inhibitors are better than the initial ‘tumour infiltrating lymphocytes (white cells)’, and it may be that CAR T-cells are better than check point inhibitors. But, what might end up happening is that each person might need a combination to get the perfect re-arming of their immune system. So it’s an ever-evolving process.
Q: How does the CAR T-cell therapy work then?
A: Look at the picture (below) of the pink and yellow cells; this is a real picture using “electron microscopy”. It shows the little ‘fingers’ of immune system cells ‘talking’ to other cells. I have spent years trying to understand how it does this … it’s like a nose and tongue ‘sniffing and tasting’ another cell and deciding “do I need to do anything here?”. Immune cells are highly connected to the environment they live in; they talk to nerve cells, sense nutrients, and respond to levels of stress hormones. CAR T-cell therapy is giving the immune cells ‘a licence to kill’. Arming the immune cell with the ability to instantly recognise the cell as cancer and assassinate it. It is no longer vulnerable. It’s like giving the immune cell the key to the cancer cell lock, but not to any other lock, and when the lock is opened the cancer cell is killed. Because of this direct arming of the immune cell, it is potentially no longer vulnerable to the cancer cell’s ability to hypnotise and confuse the immune cell into ignoring the cancer cell, but also it’s less likely to accidentally kill other cells in the body.
The amazing clinical data for CAR T-cells has come from leukaemia therapy. CD 19 is a protein that exists on leukaemia cancer cells, and this has been used to arm some CAR T-cells, giving them the identifying key for assassinating these cancer cells. This only works on a subset of cancer, but it has inspired many labs to look for new cancer keys. In our lab, we have been looking for a “master key” of solid cancers (tumours) and we have had some breakthroughs.
Q: You said the goal of Immunotherapy is to have a high target killing cancer cell rate and minimal side effects. I googled “immunotherapy side effects” and it listed some of the same symptoms as chemotherapy and radiotherapy, such as fatigue, fever, weakness, dizziness, aches, nausea, vomiting and diarrhoea, headache. Can you comment?
A: The good news is that there is no evidence yet that there will be any of the long term or types of side effects experienced with chemotherapy or radiotherapy. However, the symptoms you mentioned have been reported especially in the early trials of using CAR T-cell immunotherapy. This was due to the unprecedented successful “killing rate” of the therapy against the blood cancer cells! The early patients experienced symptoms of a “cytokine storm” which is the immune system’s reaction to infection. The immune system was dumped with so many dead cancer cells that it responded to it as a threat! So, the immunotherapists learnt very quickly to either clinically manage those symptoms, and/or take a gentler approach in administering the therapy to avoid that immune response.
Q: At what stage is CAR T-cell therapy research at currently? When will it be available?
A: Check point inhibitor therapies and CAR T-cells against blood cancer therapies are being used now. The CAR T-cells therapy has had up to a 98% disease-free rate for some leukaemias, which is very promising. In describing our research, I like the analogy of a “lock and key” and in the last 2 years we went looking for the lock and key that would work on multiple different solid (tumour) cancers. We built our little ‘lock and key’ CAR T-cell and it first worked on breast cancer. I thought “that’s a fluke”, then it worked on another type, and by the time we got to the third, we realised we have something very promising!
In the lab, using human cells in the flask (lab conditions) our ‘key’ has been fitting the ‘locks’ of up to 9 cancer types including triple negative breast, prostate, pancreas, ovary, glioma, neuroblastoma (two very difficult to treat brain cancers). But, this is a very controlled setting (in the lab) unlike when the cancer cells are growing in a human being. To use the lock and key analogy, we can test the ‘key’ presenting only one lock in the lab, but in a human, there might be 4000 doors on a dark street that the immune T-cell is navigating to find the right door lock. But … when it then worked in the pre-clinical mouse models as a next step, that was another “Aha!” moment. We have set ourselves a very ambitious goal to hold safety studies by end of 2020.
A Phase 1 Clinical Trial is a toxicity study using small numbers of people. This is the first step in learning if the therapy is safe to administer, and if that succeeds, we will then proceed to a Phase 2 Study of Efficacy. We are looking to partner with people with expertise and existing processes to do this study. If it is effective, conservatively it would take another 5-10 years for the treatment to be available for use on humans.
Q: Can the patient use the complementary therapies at the same time as Immunotherapy?
A: Absolutely! My personal view is “Do anything that helps you to feel better, as long as it does not interfere with the therapy”. There is more than one way to help someone feel better as well as to get better. Your immune system is absolutely listening to your body, how you are feeling. If you are calmer, you are not making as many stress hormones and so not distracting your T-cells with something else.
Q: You will be engaged with the Cancer Care Centre (CCC) and community as part of your ongoing work to develop immunotherapy. What’s that about?
A: By partnering with CCC, I have several goals …
- To share new treatment innovations as they emerge
- To learn from consumers about what they want from cancer researchers, and how we can include that in our research communications
- To determine what influences there are on consumers when deciding about treatment options
Thank you Prof. Simon Barry, we look forward to hearing more from you about the progress of immunotherapy in our next edition of Healthy Living. All the best for your developing immunotherapy treatment.
Watch out for part 2 of Talking with Prof. Simon Barry!
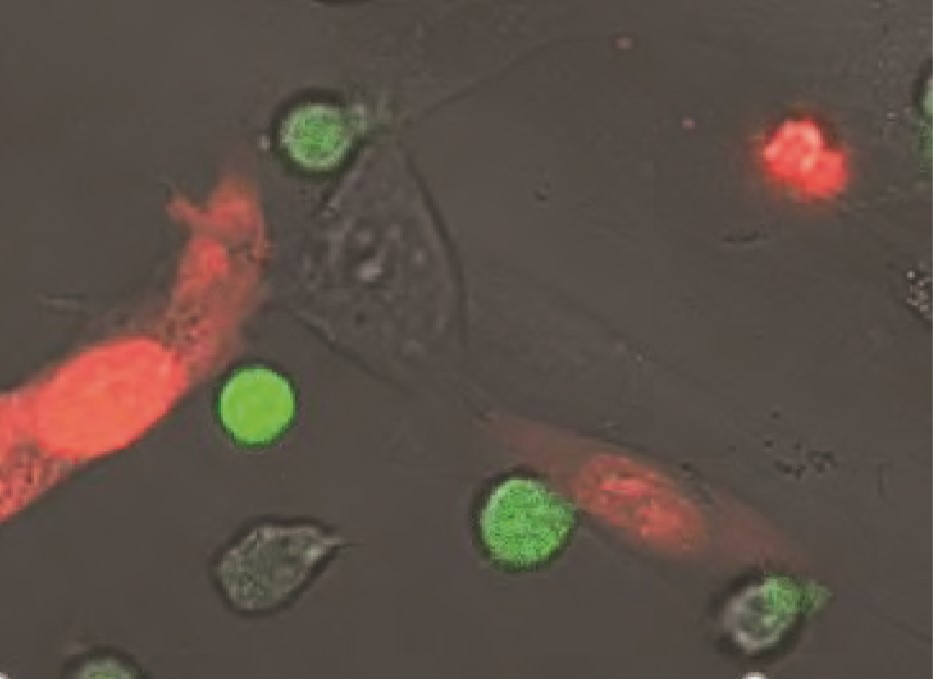 Left: This is a shot from an assay in the lab where we test whether the CAR T-cells can kill the cancer cells – the cancer cells go red when they have been assassinated
Left: This is a shot from an assay in the lab where we test whether the CAR T-cells can kill the cancer cells – the cancer cells go red when they have been assassinated
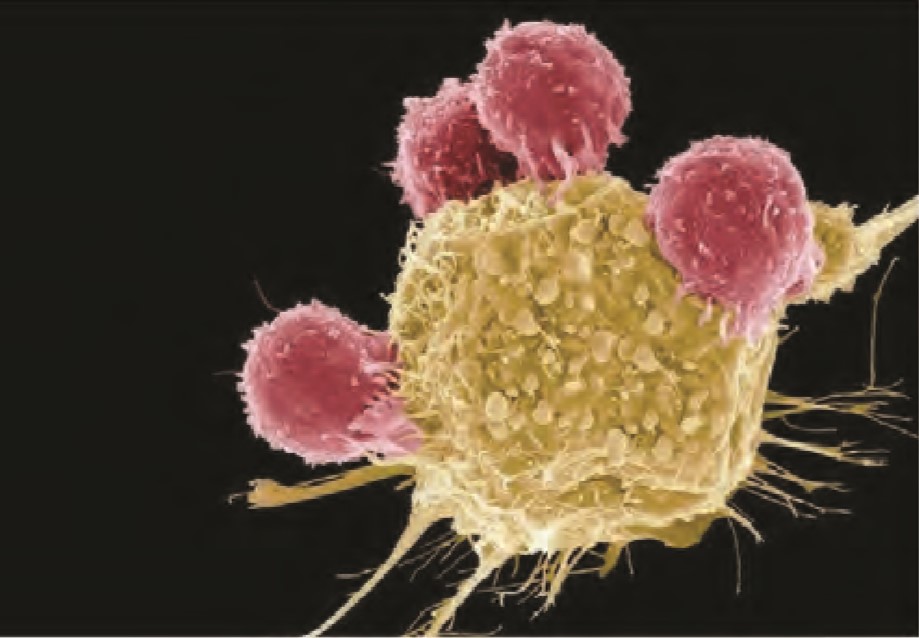 Right: This is a photo of “Immune soldiers” – immune cell (pink) reaching out and sniffing out the cancer cell (yellow)
Right: This is a photo of “Immune soldiers” – immune cell (pink) reaching out and sniffing out the cancer cell (yellow)

No comments for this article.
The comments are closed.